2023
|
Osma-Garcia, Ines C.; Mouysset, Mailys; Capitan-Sobrino, Dunja; Aubert, Yann; Turner, Martin; Diaz-Muñoz, Manuel D. The RNA binding proteins TIA1 and TIAL1 promote Mcl1 mRNA translation to protect germinal center responses from apoptosis Journal Article In: Cellular & Molecular Immunology, 2023, ISSN: 2042-0226. @article{Osma-Garcia2023b,
title = {The RNA binding proteins TIA1 and TIAL1 promote Mcl1 mRNA translation to protect germinal center responses from apoptosis},
author = {Osma-Garcia, Ines C.
and Mouysset, Mailys
and Capitan-Sobrino, Dunja
and Aubert, Yann
and Turner, Martin
and Diaz-Mu{ñ}oz, Manuel D.},
url = {https://doi.org/10.1038/s41423-023-01063-4},
doi = {10.1038/s41423-023-01063-4},
issn = {2042-0226},
year = {2023},
date = {2023-07-20},
urldate = {2023-07-20},
journal = {Cellular & Molecular Immunology},
abstract = {Germinal centers (GCs) are essential for the establishment of long-lasting antibody responses. GC B cells rely on post-transcriptional RNA mechanisms to translate activation-associated transcriptional programs into functional changes in the cell proteome. However, the critical proteins driving these key mechanisms are still unknown. Here, we show that the RNA binding proteins TIA1 and TIAL1 are required for the generation of long-lasting GC responses. TIA1- and TIAL1-deficient GC B cells fail to undergo antigen-mediated positive selection, expansion and differentiation into B-cell clones producing high-affinity antibodies. Mechanistically, TIA1 and TIAL1 control the transcriptional identity of dark- and light-zone GC B cells and enable timely expression of the prosurvival molecule MCL1. Thus, we demonstrate here that TIA1 and TIAL1 are key players in the post-transcriptional program that selects high-affinity antigen-specific GC B cells.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Germinal centers (GCs) are essential for the establishment of long-lasting antibody responses. GC B cells rely on post-transcriptional RNA mechanisms to translate activation-associated transcriptional programs into functional changes in the cell proteome. However, the critical proteins driving these key mechanisms are still unknown. Here, we show that the RNA binding proteins TIA1 and TIAL1 are required for the generation of long-lasting GC responses. TIA1- and TIAL1-deficient GC B cells fail to undergo antigen-mediated positive selection, expansion and differentiation into B-cell clones producing high-affinity antibodies. Mechanistically, TIA1 and TIAL1 control the transcriptional identity of dark- and light-zone GC B cells and enable timely expression of the prosurvival molecule MCL1. Thus, we demonstrate here that TIA1 and TIAL1 are key players in the post-transcriptional program that selects high-affinity antigen-specific GC B cells. |
2022
|
Matheson, Louise S.; Petkau, Georg; S'aenz-Narciso, Beatriz; D'Angeli, Vanessa; McHugh, Jessica; Newman, Rebecca; Munford, Haydn; West, James; Chakraborty, Krishnendu; Roberts, Jennie; Łukasiak, Sebastian; D'iaz-Muñoz, Manuel D.; Bell, Sarah E.; Dimeloe, Sarah; Turner, Martin Multiomics analysis couples mRNA turnover and translational control of glutamine metabolism to the differentiation of the activated CD4+ T cell Journal Article In: Scientific Reports, vol. 12, no. 1, pp. 19657, 2022, ISSN: 2045-2322. @article{Matheson2022,
title = {Multiomics analysis couples mRNA turnover and translational control of glutamine metabolism to the differentiation of the activated CD4+ T cell},
author = {Matheson, Louise S.
and Petkau, Georg
and S{'a}enz-Narciso, Beatriz
and D'Angeli, Vanessa
and McHugh, Jessica
and Newman, Rebecca
and Munford, Haydn
and West, James
and Chakraborty, Krishnendu
and Roberts, Jennie
and {Ł}ukasiak, Sebastian
and D{'i}az-Mu{ñ}oz, Manuel D.
and Bell, Sarah E.
and Dimeloe, Sarah
and Turner, Martin},
url = {https://doi.org/10.1038/s41598-022-24132-6},
doi = {10.1038/s41598-022-24132-6},
issn = {2045-2322},
year = {2022},
date = {2022-11-16},
journal = {Scientific Reports},
volume = {12},
number = {1},
pages = {19657},
abstract = {The ZFP36 family of RNA-binding proteins acts post-transcriptionally to repress translation and promote RNA decay. Studies of genes and pathways regulated by the ZFP36 family in CD4+ T cells have focussed largely on cytokines, but their impact on metabolic reprogramming and differentiation is unclear. Using CD4+ T cells lacking Zfp36 and Zfp36l1, we combined the quantification of mRNA transcription, stability, abundance and translation with crosslinking immunoprecipitation and metabolic profiling to determine how they regulate T cell metabolism and differentiation. Our results suggest that ZFP36 and ZFP36L1 act directly to limit the expression of genes driving anabolic processes by two distinct routes: by targeting transcription factors and by targeting transcripts encoding rate-limiting enzymes. These enzymes span numerous metabolic pathways including glycolysis, one-carbon metabolism and glutaminolysis. Direct binding and repression of transcripts encoding glutamine transporter SLC38A2 correlated with increased cellular glutamine content in ZFP36/ZFP36L1-deficient T cells. Increased conversion of glutamine to $alpha$-ketoglutarate in these cells was consistent with direct binding of ZFP36/ZFP36L1 to Gls (encoding glutaminase) and Glud1 (encoding glutamate dehydrogenase). We propose that ZFP36 and ZFP36L1 as well as glutamine and $alpha$-ketoglutarate are limiting factors for the acquisition of the cytotoxic CD4+ T cell fate. Our data implicate ZFP36 and ZFP36L1 in limiting glutamine anaplerosis and differentiation of activated CD4+ T cells, likely mediated by direct binding to transcripts of critical genes that drive these processes.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
The ZFP36 family of RNA-binding proteins acts post-transcriptionally to repress translation and promote RNA decay. Studies of genes and pathways regulated by the ZFP36 family in CD4+ T cells have focussed largely on cytokines, but their impact on metabolic reprogramming and differentiation is unclear. Using CD4+ T cells lacking Zfp36 and Zfp36l1, we combined the quantification of mRNA transcription, stability, abundance and translation with crosslinking immunoprecipitation and metabolic profiling to determine how they regulate T cell metabolism and differentiation. Our results suggest that ZFP36 and ZFP36L1 act directly to limit the expression of genes driving anabolic processes by two distinct routes: by targeting transcription factors and by targeting transcripts encoding rate-limiting enzymes. These enzymes span numerous metabolic pathways including glycolysis, one-carbon metabolism and glutaminolysis. Direct binding and repression of transcripts encoding glutamine transporter SLC38A2 correlated with increased cellular glutamine content in ZFP36/ZFP36L1-deficient T cells. Increased conversion of glutamine to $alpha$-ketoglutarate in these cells was consistent with direct binding of ZFP36/ZFP36L1 to Gls (encoding glutaminase) and Glud1 (encoding glutamate dehydrogenase). We propose that ZFP36 and ZFP36L1 as well as glutamine and $alpha$-ketoglutarate are limiting factors for the acquisition of the cytotoxic CD4+ T cell fate. Our data implicate ZFP36 and ZFP36L1 in limiting glutamine anaplerosis and differentiation of activated CD4+ T cells, likely mediated by direct binding to transcripts of critical genes that drive these processes. |
Diaz-Munoz, M. D.; Osma-Garcia, I. C. The RNA regulatory programs that govern lymphocyte development and function Journal Article In: Wiley Interdiscip Rev RNA, vol. 13, no. 1, pp. e1683, 2022, ISSN: 1757-7012 (Electronic)
1757-7004 (Linking). @article{RN3b,
title = {The RNA regulatory programs that govern lymphocyte development and function},
author = {Diaz-Munoz, M. D. and Osma-Garcia, I. C.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/34327847},
doi = {10.1002/wrna.1683},
issn = {1757-7012 (Electronic)
1757-7004 (Linking)},
year = {2022},
date = {2022-01-01},
journal = {Wiley Interdiscip Rev RNA},
volume = {13},
number = {1},
pages = {e1683},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Cacheiro-Llaguno, Cristina; Hernández-Subirá, Elena; Díaz-Muñoz, Manuel D.; Fresno, Manuel; Serrador, Juan M.; Íñiguez, Miguel A. Regulation of Cyclooxygenase-2 Expression in Human T Cells by Glucocorticoid Receptor-Mediated Transrepression of Nuclear Factor of Activated T Cells Journal Article In: International Journal of Molecular Sciences, vol. 23, no. 21, 2022, ISSN: 1422-0067. @article{ijms232113275,
title = {Regulation of Cyclooxygenase-2 Expression in Human T Cells by Glucocorticoid Receptor-Mediated Transrepression of Nuclear Factor of Activated T Cells},
author = {Cacheiro-Llaguno, Cristina and Hernández-Subirá, Elena and Díaz-Muñoz, Manuel D. and Fresno, Manuel and Serrador, Juan M. and Íñiguez, Miguel A.},
url = {https://www.mdpi.com/1422-0067/23/21/13275},
doi = {10.3390/ijms232113275},
issn = {1422-0067},
year = {2022},
date = {2022-01-01},
journal = {International Journal of Molecular Sciences},
volume = {23},
number = {21},
abstract = {Cyclooxygenase (COX) is the key enzyme in prostanoid synthesis from arachidonic acid (AA). Two isoforms, named COX-1 and COX-2, are expressed in mammalian tissues. The expression of COX-2 isoform is induced by several stimuli including cytokines and mitogens, and this induction is inhibited by glucocorticoids (GCs). We have previously shown that the transcriptional induction of COX-2 occurs early after T cell receptor (TCR) triggering, suggesting functional implications of this enzyme in T cell activation. Here, we show that dexamethasone (Dex) inhibits nuclear factor of activated T cells (NFAT)-mediated COX-2 transcriptional induction upon T cell activation. This effect is dependent on the presence of the GC receptor (GR), but independent of a functional DNA binding domain, as the activation-deficient GRLS7 mutant was as effective as the wild-type GR in the repression of NFAT-dependent transcription. Dex treatment did not disturb NFAT dephosphorylation, but interfered with activation mediated by the N-terminal transactivation domain (TAD) of NFAT, thus pointing to a negative cross-talk between GR and NFAT at the nuclear level. These results unveil the ability of GCs to interfere with NFAT activation and the induction of pro-inflammatory genes such as COX-2, and explain some of their immunomodulatory properties in activated human T cells.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Cyclooxygenase (COX) is the key enzyme in prostanoid synthesis from arachidonic acid (AA). Two isoforms, named COX-1 and COX-2, are expressed in mammalian tissues. The expression of COX-2 isoform is induced by several stimuli including cytokines and mitogens, and this induction is inhibited by glucocorticoids (GCs). We have previously shown that the transcriptional induction of COX-2 occurs early after T cell receptor (TCR) triggering, suggesting functional implications of this enzyme in T cell activation. Here, we show that dexamethasone (Dex) inhibits nuclear factor of activated T cells (NFAT)-mediated COX-2 transcriptional induction upon T cell activation. This effect is dependent on the presence of the GC receptor (GR), but independent of a functional DNA binding domain, as the activation-deficient GRLS7 mutant was as effective as the wild-type GR in the repression of NFAT-dependent transcription. Dex treatment did not disturb NFAT dephosphorylation, but interfered with activation mediated by the N-terminal transactivation domain (TAD) of NFAT, thus pointing to a negative cross-talk between GR and NFAT at the nuclear level. These results unveil the ability of GCs to interfere with NFAT activation and the induction of pro-inflammatory genes such as COX-2, and explain some of their immunomodulatory properties in activated human T cells. |
Osma-Garcia, Ines C.; Capitan-Sobrino, Dunja; Mouysset, Mailys; Aubert, Yann; Maloudi, Orlane; Turner, Martin; Diaz-Muñoz, Manuel D. The splicing regulators TIA1 and TIAL1 are required for the expression of the DNA damage repair machinery during B cell lymphopoiesis Journal Article In: Cell Reports, vol. 41, no. 12, pp. 111869, 2022, ISSN: 2211-1247. @article{OSMAGARCIA2022111869b,
title = {The splicing regulators TIA1 and TIAL1 are required for the expression of the DNA damage repair machinery during B cell lymphopoiesis},
author = {Ines C. Osma-Garcia and Dunja Capitan-Sobrino and Mailys Mouysset and Yann Aubert and Orlane Maloudi and Martin Turner and Manuel D. Diaz-Muñoz},
url = {https://www.sciencedirect.com/science/article/pii/S221112472201765X},
doi = {https://doi.org/10.1016/j.celrep.2022.111869},
issn = {2211-1247},
year = {2022},
date = {2022-01-01},
journal = {Cell Reports},
volume = {41},
number = {12},
pages = {111869},
abstract = {Summary
B cell lymphopoiesis requires dynamic modulation of the B cell transcriptome for timely coordination of somatic mutagenesis and DNA repair in progenitor B (pro-B) cells. Here, we show that, in pro-B cells, the RNA-binding proteins T cell intracellular antigen 1 (TIA1) and TIA1-like protein (TIAL1) act redundantly to enable developmental progression. They are global splicing regulators that control the expression of hundreds of mRNAs, including those involved in DNA damage repair. Mechanistically, TIA1 and TIAL1 bind to 5′ splice sites for exon definition, splicing, and expression of DNA damage sensors, such as Chek2 and Rif1. In their absence, pro-B cells show exacerbated DNA damage, altered P53 expression, and increased cell death. Our study uncovers the importance of tight regulation of RNA splicing by TIA1 and TIAL1 for the expression of integrative transcriptional programs that control DNA damage sensing and repair during B cell development.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Summary
B cell lymphopoiesis requires dynamic modulation of the B cell transcriptome for timely coordination of somatic mutagenesis and DNA repair in progenitor B (pro-B) cells. Here, we show that, in pro-B cells, the RNA-binding proteins T cell intracellular antigen 1 (TIA1) and TIA1-like protein (TIAL1) act redundantly to enable developmental progression. They are global splicing regulators that control the expression of hundreds of mRNAs, including those involved in DNA damage repair. Mechanistically, TIA1 and TIAL1 bind to 5′ splice sites for exon definition, splicing, and expression of DNA damage sensors, such as Chek2 and Rif1. In their absence, pro-B cells show exacerbated DNA damage, altered P53 expression, and increased cell death. Our study uncovers the importance of tight regulation of RNA splicing by TIA1 and TIAL1 for the expression of integrative transcriptional programs that control DNA damage sensing and repair during B cell development. |
Hu, F.; Lu, J.; Matheson, L. S.; Diaz-Munoz, M. D.; Saveliev, A.; Turner, M. Correction to: ORFLine: a bioinformatic pipeline to prioritize small open reading frames identifies candidate secreted small proteins from lymphocytes Journal Article In: Bioinformatics, 2022, ISSN: 1367-4811 (Electronic)
1367-4803 (Linking). @article{RN1b,
title = {Correction to: ORFLine: a bioinformatic pipeline to prioritize small open reading frames identifies candidate secreted small proteins from lymphocytes},
author = {Hu, F. and Lu, J. and Matheson, L. S. and Diaz-Munoz, M. D. and Saveliev, A. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/35349648},
doi = {10.1093/bioinformatics/btac162},
issn = {1367-4811 (Electronic)
1367-4803 (Linking)},
year = {2022},
date = {2022-01-01},
journal = {Bioinformatics},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2021
|
Belot, A.; Gourbeyre, O.; Palin, A.; Rubio, A.; Largounez, A.; Besson-Fournier, C.; Latour, C.; Lorgouilloux, M.; Gallitz, I.; Montagner, A.; Polizzi, A.; Regnier, M.; Smati, S.; Zhang, A. S.; Diaz-Munoz, M. D.; Steinbicker, A. U.; Guillou, H.; Roth, M. P.; Coppin, H.; Meynard, D. Endoplasmic reticulum stress controls iron metabolism through TMPRSS6 repression and hepcidin mRNA stabilization by RNA-binding protein HuR Journal Article In: Haematologica, vol. 106, no. 4, pp. 1202-1206, 2021, ISSN: 1592-8721 (Electronic)
0390-6078 (Linking). @article{RN6b,
title = {Endoplasmic reticulum stress controls iron metabolism through TMPRSS6 repression and hepcidin mRNA stabilization by RNA-binding protein HuR},
author = {Belot, A. and Gourbeyre, O. and Palin, A. and Rubio, A. and Largounez, A. and Besson-Fournier, C. and Latour, C. and Lorgouilloux, M. and Gallitz, I. and Montagner, A. and Polizzi, A. and Regnier, M. and Smati, S. and Zhang, A. S. and Diaz-Munoz, M. D. and Steinbicker, A. U. and Guillou, H. and Roth, M. P. and Coppin, H. and Meynard, D.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/32703788},
doi = {10.3324/haematol.2019.237321},
issn = {1592-8721 (Electronic)
0390-6078 (Linking)},
year = {2021},
date = {2021-01-01},
journal = {Haematologica},
volume = {106},
number = {4},
pages = {1202-1206},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Osma-Garcia, I. C.; Capitan-Sobrino, D.; Mouysset, M.; Bell, S. E.; Lebeurrier, M.; Turner, M.; Diaz-Munoz, M. D. The RNA-binding protein HuR is required for maintenance of the germinal centre response Journal Article In: Nat Commun, vol. 12, no. 1, pp. 6556, 2021, ISSN: 2041-1723 (Electronic) 2041-1723 (Linking). @article{RN2,
title = {The RNA-binding protein HuR is required for maintenance of the germinal centre response},
author = {Osma-Garcia, I. C. and Capitan-Sobrino, D. and Mouysset, M. and Bell, S. E. and Lebeurrier, M. and Turner, M. and Diaz-Munoz, M. D.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/34772950},
doi = {10.1038/s41467-021-26908-2},
issn = {2041-1723 (Electronic) 2041-1723 (Linking)},
year = {2021},
date = {2021-01-01},
journal = {Nat Commun},
volume = {12},
number = {1},
pages = {6556},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Mieulet, V.; Garnier, C.; Kieffer, Y.; Guilbert, T.; Nemati, F.; Marangoni, E.; Renault, G.; Chamming's, F.; Vincent-Salomon, A.; Mechta-Grigoriou, F. Stiffness increases with myofibroblast content and collagen density in mesenchymal high grade serous ovarian cancer Journal Article In: Sci Rep, vol. 11, no. 1, pp. 4219, 2021, ISSN: 2045-2322 (Electronic)
2045-2322 (Linking). @article{RN30b,
title = {Stiffness increases with myofibroblast content and collagen density in mesenchymal high grade serous ovarian cancer},
author = {Mieulet, V. and Garnier, C. and Kieffer, Y. and Guilbert, T. and Nemati, F. and Marangoni, E. and Renault, G. and Chamming's, F. and Vincent-Salomon, A. and Mechta-Grigoriou, F.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/33603134},
doi = {10.1038/s41598-021-83685-0},
issn = {2045-2322 (Electronic)
2045-2322 (Linking)},
year = {2021},
date = {2021-01-01},
journal = {Sci Rep},
volume = {11},
number = {1},
pages = {4219},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Hu, F.; Lu, J.; Matheson, L. S.; Diaz-Munoz, M. D.; Saveliev, A.; Turner, M. ORFLine: a bioinformatic pipeline to prioritise small open reading frames identifies candidate secreted small proteins from lymphocytes Journal Article In: Bioinformatics, 2021, ISSN: 1367-4811 (Electronic)
1367-4803 (Linking). @article{RN4b,
title = {ORFLine: a bioinformatic pipeline to prioritise small open reading frames identifies candidate secreted small proteins from lymphocytes},
author = {Hu, F. and Lu, J. and Matheson, L. S. and Diaz-Munoz, M. D. and Saveliev, A. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/33970232},
doi = {10.1093/bioinformatics/btab339},
issn = {1367-4811 (Electronic)
1367-4803 (Linking)},
year = {2021},
date = {2021-01-01},
journal = {Bioinformatics},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Boccasavia, V. L.; Bovolenta, E. R.; Villanueva, A.; Borroto, A.; Oeste, C. L.; van Santen, H. M.; Prieto, C.; Alonso-Lopez, D.; Diaz-Munoz, M. D.; Batista, F. D.; Alarcon, B. Antigen presentation between T cells drives Th17 polarization under conditions of limiting antigen Journal Article In: Cell Rep, vol. 34, no. 11, pp. 108861, 2021, ISSN: 2211-1247 (Electronic). @article{RN5b,
title = {Antigen presentation between T cells drives Th17 polarization under conditions of limiting antigen},
author = {Boccasavia, V. L. and Bovolenta, E. R. and Villanueva, A. and Borroto, A. and Oeste, C. L. and van Santen, H. M. and Prieto, C. and Alonso-Lopez, D. and Diaz-Munoz, M. D. and Batista, F. D. and Alarcon, B.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/33730591},
doi = {10.1016/j.celrep.2021.108861},
issn = {2211-1247 (Electronic)},
year = {2021},
date = {2021-01-01},
journal = {Cell Rep},
volume = {34},
number = {11},
pages = {108861},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2020
|
Lee, S.; Micalizzi, D.; Truesdell, S. S.; Bukhari, S. I. A.; Boukhali, M.; Lombardi-Story, J.; Kato, Y.; Choo, M. K.; Dey-Guha, I.; Ji, F.; Nicholson, B. T.; Myers, D. T.; Lee, D.; Mazzola, M. A.; Raheja, R.; Langenbucher, A.; Haradhvala, N. J.; Lawrence, M. S.; Gandhi, R.; Tiedje, C.; Diaz-Munoz, M. D.; Sweetser, D. A.; Sadreyev, R.; Sykes, D.; Haas, W.; Haber, D. A.; Maheswaran, S.; Vasudevan, S. A post-transcriptional program of chemoresistance by AU-rich elements and TTP in quiescent leukemic cells Journal Article In: Genome Biol, vol. 21, no. 1, pp. 33, 2020, ISSN: 1474-760X (Electronic)
1474-7596 (Linking). @article{RN7b,
title = {A post-transcriptional program of chemoresistance by AU-rich elements and TTP in quiescent leukemic cells},
author = {Lee, S. and Micalizzi, D. and Truesdell, S. S. and Bukhari, S. I. A. and Boukhali, M. and Lombardi-Story, J. and Kato, Y. and Choo, M. K. and Dey-Guha, I. and Ji, F. and Nicholson, B. T. and Myers, D. T. and Lee, D. and Mazzola, M. A. and Raheja, R. and Langenbucher, A. and Haradhvala, N. J. and Lawrence, M. S. and Gandhi, R. and Tiedje, C. and Diaz-Munoz, M. D. and Sweetser, D. A. and Sadreyev, R. and Sykes, D. and Haas, W. and Haber, D. A. and Maheswaran, S. and Vasudevan, S.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/32039742},
doi = {10.1186/s13059-020-1936-4},
issn = {1474-760X (Electronic)
1474-7596 (Linking)},
year = {2020},
date = {2020-01-01},
journal = {Genome Biol},
volume = {21},
number = {1},
pages = {33},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2019
|
Gentric, G.; Kieffer, Y.; Mieulet, V.; Goundiam, O.; Bonneau, C.; Nemati, F.; Hurbain, I.; Raposo, G.; Popova, T.; Stern, M. H.; Lallemand-Breitenbach, V.; Muller, S.; Caneque, T.; Rodriguez, R.; Vincent-Salomon, A.; de The, H.; Rossignol, R.; Mechta-Grigoriou, F. PML-Regulated Mitochondrial Metabolism Enhances Chemosensitivity in Human Ovarian Cancers Journal Article In: Cell Metab, vol. 29, no. 1, pp. 156-173 e10, 2019, ISSN: 1932-7420 (Electronic)
1550-4131 (Linking). @article{RN31b,
title = {PML-Regulated Mitochondrial Metabolism Enhances Chemosensitivity in Human Ovarian Cancers},
author = {Gentric, G. and Kieffer, Y. and Mieulet, V. and Goundiam, O. and Bonneau, C. and Nemati, F. and Hurbain, I. and Raposo, G. and Popova, T. and Stern, M. H. and Lallemand-Breitenbach, V. and Muller, S. and Caneque, T. and Rodriguez, R. and Vincent-Salomon, A. and de The, H. and Rossignol, R. and Mechta-Grigoriou, F.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/30244973},
doi = {10.1016/j.cmet.2018.09.002},
issn = {1932-7420 (Electronic)
1550-4131 (Linking)},
year = {2019},
date = {2019-01-01},
journal = {Cell Metab},
volume = {29},
number = {1},
pages = {156-173 e10},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Trulley, P.; Snieckute, G.; Bekker-Jensen, D.; Menon, M. B.; Freund, R.; Kotlyarov, A.; Olsen, J. V.; Diaz-Munoz, M. D.; Turner, M.; Bekker-Jensen, S.; Gaestel, M.; Tiedje, C. Alternative Translation Initiation Generates a Functionally Distinct Isoform of the Stress-Activated Protein Kinase MK2 Journal Article In: Cell Rep, vol. 27, no. 10, pp. 2859-2870 e6, 2019, ISSN: 2211-1247 (Electronic). @article{RN8b,
title = {Alternative Translation Initiation Generates a Functionally Distinct Isoform of the Stress-Activated Protein Kinase MK2},
author = {Trulley, P. and Snieckute, G. and Bekker-Jensen, D. and Menon, M. B. and Freund, R. and Kotlyarov, A. and Olsen, J. V. and Diaz-Munoz, M. D. and Turner, M. and Bekker-Jensen, S. and Gaestel, M. and Tiedje, C.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/31167133},
doi = {10.1016/j.celrep.2019.05.024},
issn = {2211-1247 (Electronic)},
year = {2019},
date = {2019-01-01},
journal = {Cell Rep},
volume = {27},
number = {10},
pages = {2859-2870 e6},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2018
|
Turner, M.; Diaz-Munoz, M. D. RNA-binding proteins control gene expression and cell fate in the immune system Journal Article In: Nat Immunol, vol. 19, no. 2, pp. 120-129, 2018, ISSN: 1529-2916 (Electronic)
1529-2908 (Linking). @article{RN12b,
title = {RNA-binding proteins control gene expression and cell fate in the immune system},
author = {Turner, M. and Diaz-Munoz, M. D.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29348497},
doi = {10.1038/s41590-017-0028-4},
issn = {1529-2916 (Electronic)
1529-2908 (Linking)},
year = {2018},
date = {2018-01-01},
journal = {Nat Immunol},
volume = {19},
number = {2},
pages = {120-129},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Monzon-Casanova, E.; Screen, M.; Diaz-Munoz, M. D.; Coulson, R. M. R.; Bell, S. E.; Lamers, G.; Solimena, M.; Smith, C. W. J.; Turner, M. The RNA-binding protein PTBP1 is necessary for B cell selection in germinal centers Journal Article In: Nat Immunol, vol. 19, no. 3, pp. 267-278, 2018, ISSN: 1529-2916 (Electronic)
1529-2908 (Linking). @article{RN11b,
title = {The RNA-binding protein PTBP1 is necessary for B cell selection in germinal centers},
author = {Monzon-Casanova, E. and Screen, M. and Diaz-Munoz, M. D. and Coulson, R. M. R. and Bell, S. E. and Lamers, G. and Solimena, M. and Smith, C. W. J. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29358707},
doi = {10.1038/s41590-017-0035-5},
issn = {1529-2916 (Electronic)
1529-2908 (Linking)},
year = {2018},
date = {2018-01-01},
journal = {Nat Immunol},
volume = {19},
number = {3},
pages = {267-278},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Mendoza, P.; Martinez-Martin, N.; Bovolenta, E. R.; Reyes-Garau, D.; Hernansanz-Agustin, P.; Delgado, P.; Diaz-Munoz, M. D.; Oeste, C. L.; Fernandez-Pisonero, I.; Castellano, E.; Martinez-Ruiz, A.; Alonso-Lopez, D.; Santos, E.; Bustelo, X. R.; Kurosaki, T.; Alarcon, B. R-Ras2 is required for germinal center formation to aid B cells during energetically demanding processes Journal Article In: Sci Signal, vol. 11, no. 532, 2018, ISSN: 1937-9145 (Electronic)
1945-0877 (Linking). @article{RN10b,
title = {R-Ras2 is required for germinal center formation to aid B cells during energetically demanding processes},
author = {Mendoza, P. and Martinez-Martin, N. and Bovolenta, E. R. and Reyes-Garau, D. and Hernansanz-Agustin, P. and Delgado, P. and Diaz-Munoz, M. D. and Oeste, C. L. and Fernandez-Pisonero, I. and Castellano, E. and Martinez-Ruiz, A. and Alonso-Lopez, D. and Santos, E. and Bustelo, X. R. and Kurosaki, T. and Alarcon, B.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29844052},
doi = {10.1126/scisignal.aal1506},
issn = {1937-9145 (Electronic)
1945-0877 (Linking)},
year = {2018},
date = {2018-01-01},
journal = {Sci Signal},
volume = {11},
number = {532},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Givel, A. M.; Kieffer, Y.; Scholer-Dahirel, A.; Sirven, P.; Cardon, M.; Pelon, F.; Magagna, I.; Gentric, G.; Costa, A.; Bonneau, C.; Mieulet, V.; Vincent-Salomon, A.; Mechta-Grigoriou, F. miR200-regulated CXCL12beta promotes fibroblast heterogeneity and immunosuppression in ovarian cancers Journal Article In: Nat Commun, vol. 9, no. 1, pp. 1056, 2018, ISSN: 2041-1723 (Electronic)
2041-1723 (Linking). @article{RN32,
title = {miR200-regulated CXCL12beta promotes fibroblast heterogeneity and immunosuppression in ovarian cancers},
author = {Givel, A. M. and Kieffer, Y. and Scholer-Dahirel, A. and Sirven, P. and Cardon, M. and Pelon, F. and Magagna, I. and Gentric, G. and Costa, A. and Bonneau, C. and Mieulet, V. and Vincent-Salomon, A. and Mechta-Grigoriou, F.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29535360},
doi = {10.1038/s41467-018-03348-z},
issn = {2041-1723 (Electronic)
2041-1723 (Linking)},
year = {2018},
date = {2018-01-01},
journal = {Nat Commun},
volume = {9},
number = {1},
pages = {1056},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Diaz-Munoz, M. D.; Turner, M. Uncovering the Role of RNA-Binding Proteins in Gene Expression in the Immune System Journal Article In: Front Immunol, vol. 9, pp. 1094, 2018, ISSN: 1664-3224 (Print)
1664-3224 (Linking). @article{RN9b,
title = {Uncovering the Role of RNA-Binding Proteins in Gene Expression in the Immune System},
author = {Diaz-Munoz, M. D. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29875770},
doi = {10.3389/fimmu.2018.01094},
issn = {1664-3224 (Print)
1664-3224 (Linking)},
year = {2018},
date = {2018-01-01},
journal = {Front Immunol},
volume = {9},
pages = {1094},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2017
|
Diaz-Munoz, M. D.; Monzon-Casanova, E.; Turner, M. Characterization of the B Cell Transcriptome Bound by RNA-Binding Proteins with iCLIP Journal Article In: Methods Mol Biol, vol. 1623, pp. 159-179, 2017, ISSN: 1940-6029 (Electronic)
1064-3745 (Linking). @article{RN14b,
title = {Characterization of the B Cell Transcriptome Bound by RNA-Binding Proteins with iCLIP},
author = {Diaz-Munoz, M. D. and Monzon-Casanova, E. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/28589356},
doi = {10.1007/978-1-4939-7095-7_14},
issn = {1940-6029 (Electronic)
1064-3745 (Linking)},
year = {2017},
date = {2017-01-01},
journal = {Methods Mol Biol},
volume = {1623},
pages = {159-179},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Diaz-Munoz, M. D.; Kiselev, V. Y.; Le Novere, N.; Curk, T.; Ule, J.; Turner, M. Tia1 dependent regulation of mRNA subcellular location and translation controls p53 expression in B cells Journal Article In: Nat Commun, vol. 8, no. 1, pp. 530, 2017, ISSN: 2041-1723 (Electronic)
2041-1723 (Linking). @article{RN13b,
title = {Tia1 dependent regulation of mRNA subcellular location and translation controls p53 expression in B cells},
author = {Diaz-Munoz, M. D. and Kiselev, V. Y. and Le Novere, N. and Curk, T. and Ule, J. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/28904350},
doi = {10.1038/s41467-017-00454-2},
issn = {2041-1723 (Electronic)
2041-1723 (Linking)},
year = {2017},
date = {2017-01-01},
journal = {Nat Commun},
volume = {8},
number = {1},
pages = {530},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2016
|
Tiedje, C.; Diaz-Munoz, M. D.; Trulley, P.; Ahlfors, H.; Laass, K.; Blackshear, P. J.; Turner, M.; Gaestel, M. The RNA-binding protein TTP is a global post-transcriptional regulator of feedback control in inflammation Journal Article In: Nucleic Acids Res, vol. 44, no. 15, pp. 7418-40, 2016, ISSN: 1362-4962 (Electronic)
0305-1048 (Linking). @article{RN15b,
title = {The RNA-binding protein TTP is a global post-transcriptional regulator of feedback control in inflammation},
author = {Tiedje, C. and Diaz-Munoz, M. D. and Trulley, P. and Ahlfors, H. and Laass, K. and Blackshear, P. J. and Turner, M. and Gaestel, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/27220464},
doi = {10.1093/nar/gkw474},
issn = {1362-4962 (Electronic)
0305-1048 (Linking)},
year = {2016},
date = {2016-01-01},
journal = {Nucleic Acids Res},
volume = {44},
number = {15},
pages = {7418-40},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Galloway, A.; Saveliev, A.; Lukasiak, S.; Hodson, D. J.; Bolland, D.; Balmanno, K.; Ahlfors, H.; Monzon-Casanova, E.; Mannurita, S. C.; Bell, L. S.; Andrews, S.; Diaz-Munoz, M. D.; Cook, S. J.; Corcoran, A.; Turner, M. RNA-binding proteins ZFP36L1 and ZFP36L2 promote cell quiescence Journal Article In: Science, vol. 352, no. 6284, pp. 453-9, 2016, ISSN: 1095-9203 (Electronic)
0036-8075 (Linking). @article{RN16b,
title = {RNA-binding proteins ZFP36L1 and ZFP36L2 promote cell quiescence},
author = {Galloway, A. and Saveliev, A. and Lukasiak, S. and Hodson, D. J. and Bolland, D. and Balmanno, K. and Ahlfors, H. and Monzon-Casanova, E. and Mannurita, S. C. and Bell, L. S. and Andrews, S. and Diaz-Munoz, M. D. and Cook, S. J. and Corcoran, A. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/27102483},
doi = {10.1126/science.aad5978},
issn = {1095-9203 (Electronic)
0036-8075 (Linking)},
year = {2016},
date = {2016-01-01},
journal = {Science},
volume = {352},
number = {6284},
pages = {453-9},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2015
|
Diaz-Munoz, M. D.; Bell, S. E.; Turner, M. Deletion of AU-rich elements within the Bcl2 3'UTR reduces protein expression and B cell survival in vivo Journal Article In: PLoS One, vol. 10, no. 2, pp. e0116899, 2015, ISSN: 1932-6203 (Electronic)
1932-6203 (Linking). @article{RN20b,
title = {Deletion of AU-rich elements within the Bcl2 3'UTR reduces protein expression and B cell survival in vivo},
author = {Diaz-Munoz, M. D. and Bell, S. E. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/25680182},
doi = {10.1371/journal.pone.0116899},
issn = {1932-6203 (Electronic)
1932-6203 (Linking)},
year = {2015},
date = {2015-01-01},
journal = {PLoS One},
volume = {10},
number = {2},
pages = {e0116899},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Diaz-Munoz, M. D.; Bell, S. E.; Fairfax, K.; Monzon-Casanova, E.; Cunningham, A. F.; Gonzalez-Porta, M.; Andrews, S. R.; Bunik, V. I.; Zarnack, K.; Curk, T.; Heggermont, W. A.; Heymans, S.; Gibson, G. E.; Kontoyiannis, D. L.; Ule, J.; Turner, M. The RNA-binding protein HuR is essential for the B cell antibody response Journal Article In: Nat Immunol, vol. 16, no. 4, pp. 415-25, 2015, ISSN: 1529-2916 (Electronic)
1529-2908 (Linking). @article{RN19b,
title = {The RNA-binding protein HuR is essential for the B cell antibody response},
author = {Diaz-Munoz, M. D. and Bell, S. E. and Fairfax, K. and Monzon-Casanova, E. and Cunningham, A. F. and Gonzalez-Porta, M. and Andrews, S. R. and Bunik, V. I. and Zarnack, K. and Curk, T. and Heggermont, W. A. and Heymans, S. and Gibson, G. E. and Kontoyiannis, D. L. and Ule, J. and Turner, M.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/25706746},
doi = {10.1038/ni.3115},
issn = {1529-2916 (Electronic)
1529-2908 (Linking)},
year = {2015},
date = {2015-01-01},
journal = {Nat Immunol},
volume = {16},
number = {4},
pages = {415-25},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
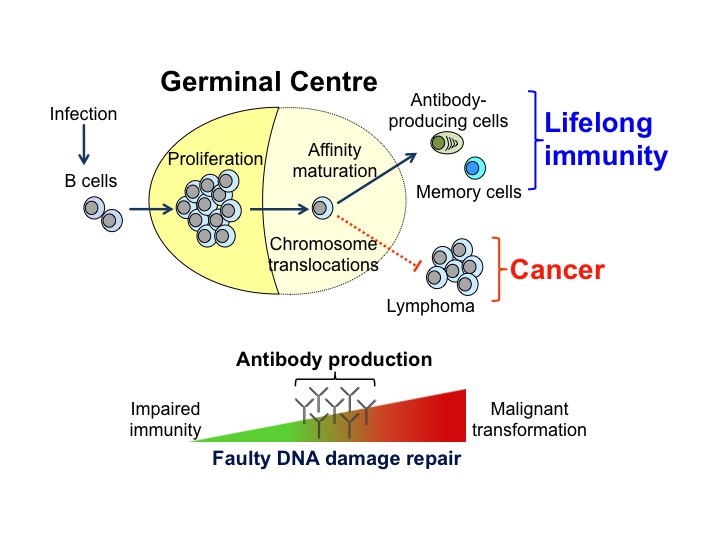 Germinal centres (GCs) are a hallmark in the fight against pathogens but they are also the source of most non- Hodgkin’s B cell lymphomas. GCs are distinctive anatomical structures formed in our secondary immune organs, such as spleen and lymph nodes, upon infection. They are not only sites for B cell clonal expansion but the places where the antibody repertoire diversifies. In GCs, B cells undergo affinity maturation. This is the process by which mutagenesis of the immunoglobulin Ig genes followed by cell selection generate antibody- producing cells and memory B cells that protect us against recurrent infections. Tight modulation of Ig somatic hypermutation and DNA damage repair is essential for antibody production, and miss- targeted mutations can lead to chromosomal translocation and B cell tumor transformation.
Germinal centres (GCs) are a hallmark in the fight against pathogens but they are also the source of most non- Hodgkin’s B cell lymphomas. GCs are distinctive anatomical structures formed in our secondary immune organs, such as spleen and lymph nodes, upon infection. They are not only sites for B cell clonal expansion but the places where the antibody repertoire diversifies. In GCs, B cells undergo affinity maturation. This is the process by which mutagenesis of the immunoglobulin Ig genes followed by cell selection generate antibody- producing cells and memory B cells that protect us against recurrent infections. Tight modulation of Ig somatic hypermutation and DNA damage repair is essential for antibody production, and miss- targeted mutations can lead to chromosomal translocation and B cell tumor transformation.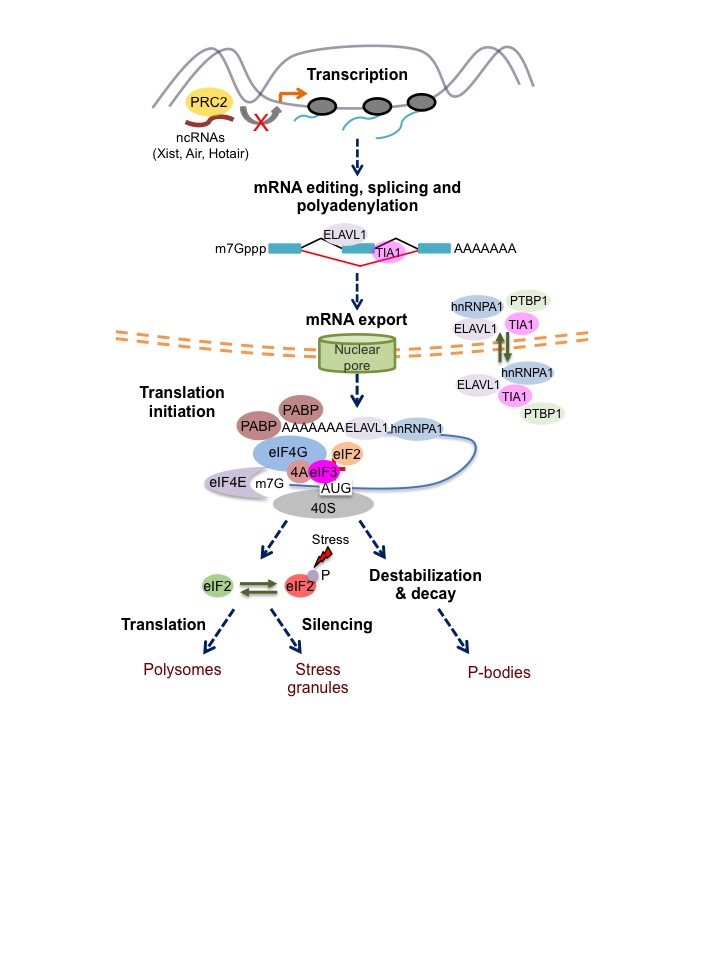 Understanding how RNA binding proteins enable Ig- class switch recombination and somatic hypermutation in GC B cells while preserving the genome integrity is fundamental in our research. Mutagenesis and DNA repair are highly regulated processes linked to the cell cycle. The existence of different cell cycle checkpoints (at G1, S and M phases) is essential for evaluation and depletion of GC B cells with extensive mutations. RNA binding proteins plays a fundamental role in these processes as they not only control lymphocyte growth and cell cycle progression but also modulate timely translation of key cell cycle checkpoint proteins and DNA damage regulators like p53.
Understanding how RNA binding proteins enable Ig- class switch recombination and somatic hypermutation in GC B cells while preserving the genome integrity is fundamental in our research. Mutagenesis and DNA repair are highly regulated processes linked to the cell cycle. The existence of different cell cycle checkpoints (at G1, S and M phases) is essential for evaluation and depletion of GC B cells with extensive mutations. RNA binding proteins plays a fundamental role in these processes as they not only control lymphocyte growth and cell cycle progression but also modulate timely translation of key cell cycle checkpoint proteins and DNA damage regulators like p53.
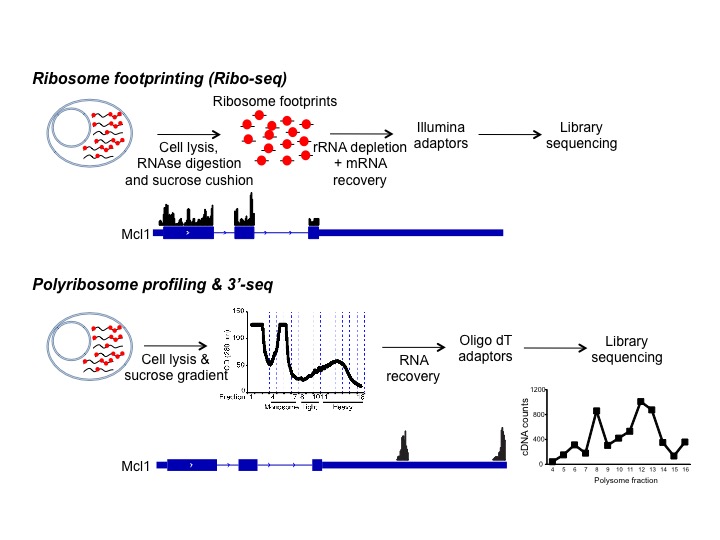 Understanding the complex regulation of lymphocyte development and differentiation requires deep analysis of the cell genome, transcriptome and translatome. The development of next-generation sequencing (NGS) has led to the definition of networks of mRNA molecules that response similarly to a given stimulus. In these early days, recent technologies are capable of both deciphering the RNA binding properties of hundreds of proteins and identify the set of RNA binding proteins associated to a single transcript, but how this knowledge is translated into regulation of lymphocyte function is under intense scrutiny in our lab.
Understanding the complex regulation of lymphocyte development and differentiation requires deep analysis of the cell genome, transcriptome and translatome. The development of next-generation sequencing (NGS) has led to the definition of networks of mRNA molecules that response similarly to a given stimulus. In these early days, recent technologies are capable of both deciphering the RNA binding properties of hundreds of proteins and identify the set of RNA binding proteins associated to a single transcript, but how this knowledge is translated into regulation of lymphocyte function is under intense scrutiny in our lab.





