2022
|
Aloulou, Meryem; Fazilleau, Nicolas The Use of Peptide-MHCII Tetramers to Identify Antigen-Specific T Follicular Helper and T Follicular Regulatory Cells Journal Article In: Methods in molecular biology (Clifton, N.J.), vol. 2380, pp. 141–147, 2022, ISSN: 1064-3745. @article{aloulou_use_2022,
title = {The Use of Peptide-MHCII Tetramers to Identify Antigen-Specific T Follicular Helper and T Follicular Regulatory Cells},
author = {Aloulou, Meryem and Fazilleau, Nicolas},
url = {https://doi.org/10.1007/978-1-0716-1736-6_12},
doi = {10.1007/978-1-0716-1736-6_12},
issn = {1064-3745},
year = {2022},
date = {2022-01-01},
journal = {Methods in molecular biology (Clifton, N.J.)},
volume = {2380},
pages = {141--147},
abstract = {Characterization and counting of the different immune cell subpopulations are largely used in order to predict the quality of vaccination or the progression of diseases. As such, flow cytometry is a valuable technology to perform an exact cartography of the immune cell subsets. In the context of B-cell responses, specialized structures emerge in B-follicles of second lymphoid organs where B-cells "undergo maturation processes under the guidance of specific T-cells, follicular helper T-cells, and follicular regulatory T-cells. Thus, tracking these cell types is of high interest, especially in the context of protein vaccination. In this purpose, we describe here, how we can track antigen-specific follicular helper T-cells and follicular regulatory T-cells by flow cytometry after protein vaccination in nonmodified wild-type mice, which ultimately provides a comprehensive way to better understand the function of these particular cells in vivo.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Characterization and counting of the different immune cell subpopulations are largely used in order to predict the quality of vaccination or the progression of diseases. As such, flow cytometry is a valuable technology to perform an exact cartography of the immune cell subsets. In the context of B-cell responses, specialized structures emerge in B-follicles of second lymphoid organs where B-cells "undergo maturation processes under the guidance of specific T-cells, follicular helper T-cells, and follicular regulatory T-cells. Thus, tracking these cell types is of high interest, especially in the context of protein vaccination. In this purpose, we describe here, how we can track antigen-specific follicular helper T-cells and follicular regulatory T-cells by flow cytometry after protein vaccination in nonmodified wild-type mice, which ultimately provides a comprehensive way to better understand the function of these particular cells in vivo. |
Ph., Kemoun; Ader, I.; Planat-Benard, V.; Dray, C.; Fazilleau, N.; Monsarrat, P.; Cousin, B.; Paupert, J.; Ousset, M.; Lorsignol, A.; Raymond-Letron, I.; Vella, B.; Valet, P.; Kirkwood, T.; Beard, J.; Pénicaud, L.; Casteilla, L. A gerophysiology perspective on healthy ageing. Journal Article In: Ageing Research Reviews, vol. 73, pp. 101537, 2022. @article{Ph.2022,
title = {A gerophysiology perspective on healthy ageing.},
author = { Kemoun Ph. and Ader, I. and Planat-Benard, V. and Dray, C. and Fazilleau, N. and Monsarrat, P. and Cousin, B. and Paupert, J. and Ousset, M. and Lorsignol, A. and Raymond-Letron, I. and Vella, B. and Valet, P. and Kirkwood, T. and Beard, J. and Pénicaud, L. and Casteilla, L.},
editor = {Elsevier Masson},
url = {https://hal.science/hal-03739142/file/Soumission%20ARR-S-21-00412-1.pdf},
doi = {10.1016/j.arr.2021.101537},
year = {2022},
date = {2022-01-01},
journal = {Ageing Research Reviews},
volume = {73},
pages = {101537},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Yang, Cui; Blaize, Gaëtan; Marrocco, Rémi; Rouquié, Nelly; Bories, Cyrielle; Gador, Mylène; Mélique, Suzanne; Joulia, Emeline; Benamar, Mehdi; Dejean, Anne S.; Daniels-Treffandier, Hélène; Love, Paul E.; Fazilleau, Nicolas; Saoudi, Abdelhadi; Lesourne, Renaud THEMIS enhances the magnitude of normal and neuroinflammatory type 1 immune responses by promoting TCR-independent signals Journal Article In: Science Signaling, vol. 15, no. 742, pp. eabl5343, 2022, (Publisher: American Association for the Advancement of Science). @article{yang_themis_2022,
title = {THEMIS enhances the magnitude of normal and neuroinflammatory type 1 immune responses by promoting TCR-independent signals},
author = {Yang, Cui and Blaize, Gaëtan and Marrocco, Rémi and Rouquié, Nelly and Bories, Cyrielle and Gador, Mylène and Mélique, Suzanne and Joulia, Emeline and Benamar, Mehdi and Dejean, Anne S. and Daniels-Treffandier, Hélène and Love, Paul E. and Fazilleau, Nicolas and Saoudi, Abdelhadi and Lesourne, Renaud},
url = {https://www.science.org/doi/abs/10.1126/scisignal.abl5343},
doi = {10.1126/scisignal.abl5343},
year = {2022},
date = {2022-01-01},
urldate = {2022-01-01},
journal = {Science Signaling},
volume = {15},
number = {742},
pages = {eabl5343},
note = {Publisher: American Association for the Advancement of Science},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2021
|
Martinez, F; Novarino, J; Mejía, J E; Fazilleau, N; Aloulou, M Ageing of T-dependent B cell responses Journal Article In: Immunology letters, 2021. @article{Martinez2021,
title = {Ageing of T-dependent B cell responses},
author = {F Martinez and J Novarino and J E Mejía and N Fazilleau and M Aloulou},
editor = {Elsevier},
url = {https://pdf.sciencedirectassets.com/271143/AIP/1-s2.0-S016524782100050X/main.pdf?X-Amz-Security-Token=IQoJb3JpZ2luX2VjEBgaCXVzLWVhc3QtMSJGMEQCIG%2BFwpqjaWVS40wshKKTU3J%2BxDLVNDIQoyx2CeQ6aMA8AiB0yvzzWva83RZl4WFM2SLAEUvuBjTimiFNcR18EhJgtCq0AwhwEAMaDDA1OTAwMzU0Njg2NSIMBY6RcnUcJj3%2BzM8JKpEDcDUBjOHbsoFcnlMU1e4XECeah2Yih%2BLJ60bJm%2BSGnsC3WnoyY7hNeLIY4XADtL0%2FZXbFgZmslofJLJeV5MBP%2BFcmNi%2BnsM%2BXDtMglW6YLwdF0mgEU6h8CXrX3MN8hb9dxLpKmKuSFekp8FzvzqoeIFnu6EGIHpA1Q1FUhrj8bZ%2BF9gebbYpZA%2B8%2FTix%2BY4yUtaDek82zXXPiTrIVjGZ7%2Fyryp6l%2F%2B21zTOMGCpfuk9EKO3UCVhmyXukbxz5BXjuCbcqOdLtSmGTuntcxeqWZN8E4iLTM%2BmNfWwExeg86fZLzSmNGkRZmmJwvs1LC967oQ3hPsfSKzVNH8o4J%2FwMzfmVbXHPGLH50Dfqa5bkxwg7x6p6JVFmQShMtz5MPtqfQQ0k6Jaj%2BxCUmp5mKfB4NcKV2O2rAYIGfla8ygellF87pDDdakSPebjpj2Ug4ebQZmhM4CS%2B%2BuKyk7cTgUK2PPZTXE8DFXgEkZ6erDjIeEgf0UkMRN1zHIr9r%2BUMDm%2BuHUt5t53LZqgcp4GZNpR8UeaEwi4HAgwY67AHY9WTpeeKjny9HrSTopZcnzPk2PFcSaL2SKIQ15e8UcbF59yhbV%2FjGzPu46h9qCGJ4tYADPTAEGBU8YbGGy5TkXZfh%2FhvoBA2bUoTbeW5B0I983zA94U7cU8kMRgC27h4gaLeGllCoHEnaudFPrI14cGeg9BtfMYRJUPK0gizhRd8B8brHNYHsz2YPc5c3nYUcHZPTUw6d6lREhRXDfnGtmZkIQQaUHdZ2LX44rOhsNInDfK4osXgsVpPGdINdb9YS2PPnJfUdCu0GJF9xry8gS0AKRgNVnX%2BuM4deYNT%2BEB99onYNOG7By7Gepw%3D%3D&X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Date=20210409T084508Z&X-Amz-SignedHeaders=host&X-Amz-Expires=300&X-Amz-Credential=ASIAQ3PHCVTYRDMRDBR5%2F20210409%2Fus-east-1%2Fs3%2Faws4_request&X-Amz-Signature=b56eec3daa725004f4b0149202965a04fef899116f616d10f739fcc47e9062a1&hash=a7d31bb1f7709cafa7a161144530e38c584b8b9fefd3ccca8482906315fcfca3&host=68042c943591013ac2b2430a89b270f6af2c76d8dfd086a07176afe7c76c2c61&pii=S016524782100050X&tid=spdf-c9aa0e26-91de-44e2-bec3-ea2bca7b2f45&sid=68c1aa39790ee6494568a54117c58816c059gxrqb&type=client},
doi = {https://doi.org/10.1016/j.imlet.2021.03.012},
year = {2021},
date = {2021-03-31},
journal = {Immunology letters},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Baumjohann, D; Fazilleau, N Antigen-dependent multistep differentiation of T follicular helper cells and its role in SARS-CoV-2 infection and vaccination. Journal Article In: Eur J Immunol., 2021. @article{Baumjohann2021,
title = {Antigen-dependent multistep differentiation of T follicular helper cells and its role in SARS-CoV-2 infection and vaccination.},
author = {D Baumjohann and N Fazilleau },
url = {https://doi.org/10.1002/eji.202049148},
doi = {10.1002/eji.202049148},
year = {2021},
date = {2021-03-31},
journal = {Eur J Immunol.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Maho-Vaillant, M; Pérals, C; Golinski, ML; Hébert, V; Caillot, F; Mignard, C; Riou, G; Petit, M; Viguier, M; Hertl, M; Boyer, O; Calbo, S; Fazilleau, N; Joly, P RITUXIMAB AND CORTICOSTEROID EFFECT ON DESMOGLEIN-SPECIFIC B-CELLS AND DESMOGLEIN-SPECIFIC T-FOLLICULAR-HELPER-CELLS IN PEMPHIGUS Journal Article In: J. Invest Dermatol , 2021. @article{Maho-Vaillant2021,
title = {RITUXIMAB AND CORTICOSTEROID EFFECT ON DESMOGLEIN-SPECIFIC B-CELLS AND DESMOGLEIN-SPECIFIC T-FOLLICULAR-HELPER-CELLS IN PEMPHIGUS},
author = {M Maho-Vaillant and C Pérals and ML Golinski and V Hébert and F Caillot and C Mignard and G Riou and M Petit and M Viguier and M Hertl and O Boyer and S Calbo and N Fazilleau and P Joly },
url = {https://www-sciencedirect-com.proxy.insermbiblio.inist.fr/science/article/pii/S0022202X21009970?via%3Dihub},
doi = { 10.1016/j.jid.2021.01.031},
year = {2021},
date = {2021-03-22},
journal = { J. Invest Dermatol },
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Webb, LMC; S, Fra-Bido; S, Innocentin; LS, Matheson; N, Attaf; A, Bignon; J, Novarino; N, Fazilleau; MA., Linterman Ageing promotes early T follicular helper cell differentiation by modulating expression of RBPJ Journal Article In: Aging Cell, 2021. @article{Webb2021,
title = {Ageing promotes early T follicular helper cell differentiation by modulating expression of RBPJ},
author = {Webb, LMC and Fra-Bido S and Innocentin S and Matheson LS and Attaf N and Bignon A and Novarino J and Fazilleau N
and Linterman MA. },
url = {https://onlinelibrary.wiley.com/doi/full/10.1111/acel.13295},
doi = {10.1111/acel.13295},
year = {2021},
date = {2021-01-02},
journal = {Aging Cell},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2020
|
G, Blaize; H, Daniels-Treffandier; M, Aloulou; N, Rouquié; C, Yang; M, Marcellin; M, Gador; M, Benamar; M, Ducatez; KD, Song; O, Burlet-Schiltz; A, Saoudi; PE, Love; N, Fazilleau; de Peredo A, Gonzalez; R., Lesourne CD5 signalosome coordinates antagonist TCR signals to control the generation of Treg cells induced by foreign antigens. Journal Article In: Proc Natl Acad Sci U S A, vol. 117, no. 23, pp. 12969-12979, 2020. @article{G2020,
title = {CD5 signalosome coordinates antagonist TCR signals to control the generation of Treg cells induced by foreign antigens. },
author = {Blaize G and Daniels-Treffandier H and Aloulou M and Rouquié N and Yang C and Marcellin M and Gador M and Benamar M and Ducatez M and Song KD and Burlet-Schiltz O and Saoudi A and Love PE and Fazilleau N and Gonzalez de Peredo A and Lesourne R.
},
url = {https://www.pnas.org/content/117/23/12969},
doi = {10.1073/pnas.1917182117. },
year = {2020},
date = {2020-06-09},
journal = {Proc Natl Acad Sci U S A},
volume = {117},
number = {23},
pages = {12969-12979},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
F, Briand; C, Heymes; L, Bonada; T, Angles; J, Charpentier; M, Branchereau; E, Brousseau; M, Quinsat; N, Fazilleau; R, Burcelin; T., Sulpice A 3-week nonalcoholic steatohepatitis mouse model shows elafibranor benefits on hepatic inflammation and cell death. Journal Article In: Clin Transl Sci, vol. 13, no. 3, pp. 529-538, 2020. @article{F2020,
title = {A 3-week nonalcoholic steatohepatitis mouse model shows elafibranor benefits on hepatic inflammation and cell death.},
author = {Briand F and Heymes C and Bonada L and Angles T and Charpentier J and Branchereau M and Brousseau E and Quinsat M and Fazilleau N and Burcelin R and Sulpice T.},
url = {https://ascpt.onlinelibrary.wiley.com/doi/full/10.1111/cts.12735},
doi = {10.1111/cts.12735.},
year = {2020},
date = {2020-05-01},
journal = {Clin Transl Sci},
volume = {13},
number = {3},
pages = {529-538},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
K, Mahiddine; C, Hassel; C, Murat; M, Girard; S., Guerder Tissue-Specific Factors Differentially Regulate the Expression of Antigen-Processing Enzymes During Dendritic Cell Ontogeny. Journal Article In: Front Immunol. , vol. 11, no. 453, 2020. @article{K2020,
title = {Tissue-Specific Factors Differentially Regulate the Expression of Antigen-Processing Enzymes During Dendritic Cell Ontogeny.},
author = {Mahiddine K and Hassel C and Murat C and Girard M and Guerder S.
},
url = {https://www.frontiersin.org/articles/10.3389/fimmu.2020.00453/full},
doi = {10.3389/fimmu.2020.00453.},
year = {2020},
date = {2020-03-31},
journal = {Front Immunol. },
volume = {11},
number = {453},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
F, Guerville; De Souto Barreto P,; I, Ader; S, Andrieu; L, Casteilla; C, Dray; N, Fazilleau; S, Guyonnet; D, Langin; R, Liblau; A, Parini; P, Valet; N, Vergnolle; Y, Rolland; B., Vellas Revisiting the Hallmarks of Aging to Identify Markers of Biological Age. Journal Article In: J Prev Alzheimers Dis. , vol. 7, no. 1, pp. 56-64, 2020. @article{F2020,
title = {Revisiting the Hallmarks of Aging to Identify Markers of Biological Age. },
author = {Guerville F and De Souto Barreto P, and Ader I and Andrieu S and Casteilla L and Dray C and Fazilleau N and Guyonnet S and Langin D and Liblau R and Parini A and Valet P and Vergnolle N and Rolland Y and Vellas B. },
url = {https://pubmed.ncbi.nlm.nih.gov/32010927/},
doi = {10.14283/jpad.2019.50.},
year = {2020},
date = {2020-01-01},
journal = {J Prev Alzheimers Dis. },
volume = {7},
number = {1},
pages = {56-64},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2019
|
C, Dubois; F, Martin; C, Hassel; F, Magnier; P, Daumar; C, Aubel; S, Guerder; E, Mounetou; F, Penault-Lorca; M., Bamdad Low-Dose and Long-Term Olaparib Treatment Sensitizes MDA-MB-231 and SUM1315 Triple-Negative Breast Cancers Spheroids to Fractioned Radiotherapy. Journal Article In: J Clin Med, vol. 9, no. 1, 2019. @article{C2019,
title = {Low-Dose and Long-Term Olaparib Treatment Sensitizes MDA-MB-231 and SUM1315 Triple-Negative Breast Cancers Spheroids to Fractioned Radiotherapy.},
author = {Dubois C and Martin F and Hassel C and Magnier F and Daumar P and Aubel C and Guerder S and Mounetou E and Penault-Lorca F and Bamdad M.
},
url = {https://www.mdpi.com/2077-0383/9/1/64},
doi = {10.3390/jcm9010064.},
year = {2019},
date = {2019-12-26},
journal = {J Clin Med},
volume = {9},
number = {1},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
M, Aloulou; N., Fazilleau Regulation of B cell responses by distinct populations of CD4 T cells. Journal Article In: Biomed J. , vol. 42, no. 4, pp. 243-251, 2019. @article{M2019,
title = {Regulation of B cell responses by distinct populations of CD4 T cells.},
author = {Aloulou M and Fazilleau N. },
url = {https://www.sciencedirect.com/science/article/pii/S2319417019304822},
doi = {10.1016/j.bj.2019.06.002.},
year = {2019},
date = {2019-08-01},
journal = {Biomed J. },
volume = {42},
number = {4},
pages = {243-251},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Guerder, S.; Hassel, C.; Carrier, A. Thymus-specific serine protease, a protease that shapes the CD4 T cell repertoire. Journal Article In: Immunogenetics, vol. 71, no. 3, pp. 223, 2019. @article{S2019,
title = {Thymus-specific serine protease, a protease that shapes the CD4 T cell repertoire.},
author = {Guerder, S. and Hassel, C. and Carrier, A.},
url = {https://link.springer.com/article/10.1007/s00251-018-1078-y},
doi = {10.1007/s00251-018-1078-y},
year = {2019},
date = {2019-03-01},
journal = {Immunogenetics},
volume = {71},
number = {3},
pages = {223},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2018
|
Nguyen, XH.; Dauvilliers, Y.; Quériault, C.; Pérals, C.; Romieu-Mourez, R.; Paulet, PE.; Bernard-Valnet, R.; Fazilleau, N.; Liblau, R. Circulating follicular helper T cells exhibit reduced ICOS expression and impaired function in narcolepsy type 1 patients. Journal Article In: J Autoimmun., vol. 94, pp. 134-142, 2018, ISSN: 1095-9157. @article{Nguyen2018,
title = {Circulating follicular helper T cells exhibit reduced ICOS expression and impaired function in narcolepsy type 1 patients.},
author = {Nguyen, XH. and Dauvilliers, Y. and Quériault, C. and Pérals, C. and Romieu-Mourez, R. and Paulet, PE. and Bernard-Valnet, R. and Fazilleau, N. and Liblau, R. },
url = {https://www.ncbi.nlm.nih.gov/pubmed/30104107},
doi = {10.1016/j.jaut.2018.07.021},
issn = {1095-9157},
year = {2018},
date = {2018-11-01},
journal = {J Autoimmun.},
volume = {94},
pages = {134-142},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Bernard, I.; Sacquin, A.; Kassem, S.; Benamar, M.; Colacios, C.; Gador, M.; Pérals, C.; Fazilleau, N.; Saoudi, A. A Natural Variant of the Signaling Molecule Vav1 Enhances Susceptibility to Myasthenia Gravis and Influences the T Cell Receptor Repertoire. Journal Article In: Front Immunol. , vol. 9, pp. 2399 , 2018, ISSN: 1664-3224. @article{I2018,
title = {A Natural Variant of the Signaling Molecule Vav1 Enhances Susceptibility to Myasthenia Gravis and Influences the T Cell Receptor Repertoire. },
author = {Bernard, I. and Sacquin, A. and Kassem, S. and Benamar, M. and Colacios, C. and Gador, M. and Pérals, C. and Fazilleau, N. and Saoudi, A. },
url = {https://www.ncbi.nlm.nih.gov/pubmed/30410484},
doi = {10.3389/fimmu.2018.02399},
issn = {1664-3224},
year = {2018},
date = {2018-10-25},
journal = {Front Immunol. },
volume = {9},
pages = {2399 },
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Fazilleau, N.; Aloulou, M. Several Follicular Regulatory T Cell Subsets With Distinct Phenotype and Function Emerge During Germinal Center Reactions. Journal Article In: Front Immunol. , vol. 9, pp. 1792, 2018, ISSN: 1664-3224. @article{Fazilleau2018,
title = {Several Follicular Regulatory T Cell Subsets With Distinct Phenotype and Function Emerge During Germinal Center Reactions.},
author = {Fazilleau, N. and Aloulou, M. },
url = {https://www.ncbi.nlm.nih.gov/pubmed/30150980},
doi = {10.3389/fimmu.2018.01792},
issn = {1664-3224},
year = {2018},
date = {2018-08-13},
journal = {Front Immunol. },
volume = {9},
pages = {1792},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2017
|
Asrir, A.; Aloulou, M.; Gador, M.; Perals, C.; Fazilleau, N. Interconnected subsets of memory follicular helper T cells have different effector functions Journal Article In: Nat Commun, vol. 8, no. 1, pp. 847, 2017, ISSN: 2041-1723 (Electronic) 2041-1723 (Linking). @article{RN2b,
title = {Interconnected subsets of memory follicular helper T cells have different effector functions},
author = {Asrir, A. and Aloulou, M. and Gador, M. and Perals, C. and Fazilleau, N.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/29018187},
doi = {10.1038/s41467-017-00843-7},
issn = {2041-1723 (Electronic) 2041-1723 (Linking)},
year = {2017},
date = {2017-01-01},
journal = {Nat Commun},
volume = {8},
number = {1},
pages = {847},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Serre, L.; Girard, M.; Ramadan, A.; Menut, P.; Rouquie, N.; Lucca, L. E.; Mahiddine, K.; Leobon, B.; Mars, L. T.; Guerder, S. Thymic-Specific Serine Protease Limits Central Tolerance and Exacerbates Experimental Autoimmune Encephalomyelitis Journal Article In: Journal of Immunology, vol. 199, no. 11, pp. 3748-3756, 2017, ISSN: 0022-1767. @article{RN16b,
title = {Thymic-Specific Serine Protease Limits Central Tolerance and Exacerbates Experimental Autoimmune Encephalomyelitis},
author = {Serre, L. and Girard, M. and Ramadan, A. and Menut, P. and Rouquie, N. and Lucca, L. E. and Mahiddine, K. and Leobon, B. and Mars, L. T. and Guerder, S.},
url = {<Go to ISI>://WOS:000415967800006},
doi = {10.4049/jimmunol.1700667},
issn = {0022-1767},
year = {2017},
date = {2017-01-01},
journal = {Journal of Immunology},
volume = {199},
number = {11},
pages = {3748-3756},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Sacquin, A.; Gador, M.; Fazilleau, N. The strength of BCR signaling shapes terminal development of follicular helper T cells in mice Journal Article In: Eur J Immunol, vol. 47, no. 8, pp. 1295-1304, 2017, ISSN: 1521-4141 (Electronic)
0014-2980 (Linking). @article{RN17,
title = {The strength of BCR signaling shapes terminal development of follicular helper T cells in mice},
author = {Sacquin, A. and Gador, M. and Fazilleau, N.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/28605013},
doi = {10.1002/eji.201746952},
issn = {1521-4141 (Electronic)
0014-2980 (Linking)},
year = {2017},
date = {2017-01-01},
journal = {Eur J Immunol},
volume = {47},
number = {8},
pages = {1295-1304},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2016
|
Joncker, N. T.; Bettini, S.; Boulet, D.; Guiraud, M.; Guerder, S. The site of tumor development determines immunogenicity via temporal mobilization of antigen-laden dendritic cells in draining lymph nodes Journal Article In: European Journal of Immunology, vol. 46, no. 3, pp. 609-618, 2016, ISSN: 0014-2980. @article{RN15b,
title = {The site of tumor development determines immunogenicity via temporal mobilization of antigen-laden dendritic cells in draining lymph nodes},
author = {Joncker, N. T. and Bettini, S. and Boulet, D. and Guiraud, M. and Guerder, S.},
url = {<Go to ISI>://WOS:000372353600015},
doi = {10.1002/eji.201545797},
issn = {0014-2980},
year = {2016},
date = {2016-01-01},
journal = {European Journal of Immunology},
volume = {46},
number = {3},
pages = {609-618},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Aloulou, M.; Carr, E. J.; Gador, M.; Bignon, A.; Liblau, R. S.; Fazilleau, N.; Linterman, M. A. Follicular regulatory T cells can be specific for the immunizing antigen and derive from naive T cells Journal Article In: Nat Commun, vol. 7, pp. 10579, 2016, ISSN: 2041-1723 (Electronic)
2041-1723 (Linking). @article{RN13,
title = {Follicular regulatory T cells can be specific for the immunizing antigen and derive from naive T cells},
author = {Aloulou, M. and Carr, E. J. and Gador, M. and Bignon, A. and Liblau, R. S. and Fazilleau, N. and Linterman, M. A.},
url = {http://www.ncbi.nlm.nih.gov/pubmed/26818004},
doi = {10.1038/ncomms10579},
issn = {2041-1723 (Electronic)
2041-1723 (Linking)},
year = {2016},
date = {2016-01-01},
journal = {Nat Commun},
volume = {7},
pages = {10579},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2015
|
Viret, C.; Mahiddine, K.; Baker, R. L.; Haskins, K.; Guerder, S The T Cell Repertoire-Diversifying Enzyme TSSP Contributes to Thymic Selection of Diabetogenic CD4 T Cell Specificities Reactive to ChgA and IAPP Autoantigens Journal Article In: J Immunol , vol. 5, no. 195, pp. 1964-1973, 2015. @article{Viret2015,
title = {The T Cell Repertoire-Diversifying Enzyme TSSP Contributes to Thymic Selection of Diabetogenic CD4 T Cell Specificities Reactive to ChgA and IAPP Autoantigens},
author = {Viret, C. and Mahiddine, K. and Baker, R.L. and Haskins, K. and Guerder, S },
url = {https://www.ncbi.nlm.nih.gov/pubmed/26209627},
doi = {10.4049/jimmunol.1401683},
year = {2015},
date = {2015-09-01},
journal = {J Immunol },
volume = {5},
number = {195},
pages = {1964-1973},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Serre, L.; Fazilleau, N.; Guerder, S. Central tolerance spares the private high-avidity CD4 T-cell repertoire specific for an islet antigen in NOD mice Journal Article In: Eur J Immunol, 2015, ISSN: 1521-4141 (Electronic)
0014-2980 (Linking). @article{RN3b,
title = {Central tolerance spares the private high-avidity CD4 T-cell repertoire specific for an islet antigen in NOD mice},
author = {Serre, L. and Fazilleau, N. and Guerder, S.},
url = {http://www.ncbi.nlm.nih.gov/pubmed/25884569},
doi = {10.1002/eji.201445290},
issn = {1521-4141 (Electronic)
0014-2980 (Linking)},
year = {2015},
date = {2015-01-01},
journal = {Eur J Immunol},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Chakarov, S.; Fazilleau, N. Tracking by flow cytometry antigen-specific follicular helper T cells in wild-type animals after protein vaccination Journal Article In: Methods Mol Biol, vol. 1291, pp. 39-47, 2015, ISSN: 1940-6029 (Electronic)
1064-3745 (Linking). @article{RN6b,
title = {Tracking by flow cytometry antigen-specific follicular helper T cells in wild-type animals after protein vaccination},
author = {Chakarov, S. and Fazilleau, N.},
url = {http://www.ncbi.nlm.nih.gov/pubmed/25836300},
doi = {10.1007/978-1-4939-2498-1_4},
issn = {1940-6029 (Electronic)
1064-3745 (Linking)},
year = {2015},
date = {2015-01-01},
journal = {Methods Mol Biol},
volume = {1291},
pages = {39-47},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2014
|
Chakarov, S.; Fazilleau, N. Monocyte-derived dendritic cells promote T follicular helper cell differentiation Journal Article In: EMBO Mol Med, vol. 6, pp. 590-603, 2014, ISSN: 1757-4684 (Electronic)
1757-4676 (Linking). @article{RN5b,
title = {Monocyte-derived dendritic cells promote T follicular helper cell differentiation},
author = {Chakarov, S. and Fazilleau, N.},
url = {http://www.ncbi.nlm.nih.gov/pubmed/24737871},
doi = {10.1002/emmm.201403841},
issn = {1757-4684 (Electronic)
1757-4676 (Linking)},
year = {2014},
date = {2014-01-01},
journal = {EMBO Mol Med},
volume = {6},
pages = {590-603},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2013
|
Guerder, S.; Joncker, N.; Mahiddine, K.; Serre, L. Dendritic cells in tolerance and autoimmune diabetes Journal Article In: Curr Opin Immunol, vol. 25, no. 6, pp. 670-5, 2013, ISSN: 1879-0372 (Electronic)
0952-7915 (Linking). @article{RN7b,
title = {Dendritic cells in tolerance and autoimmune diabetes},
author = {Guerder, S. and Joncker, N. and Mahiddine, K. and Serre, L.},
url = {https://www.ncbi.nlm.nih.gov/pubmed/24168964},
doi = {10.1016/j.coi.2013.10.004},
issn = {1879-0372 (Electronic)
0952-7915 (Linking)},
year = {2013},
date = {2013-01-01},
journal = {Curr Opin Immunol},
volume = {25},
number = {6},
pages = {670-5},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2012
|
Guerder, S.; Viret, C.; Luche, H.; Ardouin, L.; Malissen, B. Differential processing of self-antigens by subsets of thymic stromal cells Journal Article In: Curr Opin Immunol, vol. 24, no. 1, pp. 99-104, 2012, ISSN: 1879-0372 (Electronic)
0952-7915 (Linking). @article{RN12,
title = {Differential processing of self-antigens by subsets of thymic stromal cells},
author = {Guerder, S. and Viret, C. and Luche, H. and Ardouin, L. and Malissen, B.},
url = {http://www.ncbi.nlm.nih.gov/pubmed/22296716},
doi = {10.1016/j.coi.2012.01.008},
issn = {1879-0372 (Electronic)
0952-7915 (Linking)},
year = {2012},
date = {2012-01-01},
journal = {Curr Opin Immunol},
volume = {24},
number = {1},
pages = {99-104},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
2011
|
Viret, C.; Leung-Theung-Long, S.; Serre, L.; C.and Vignali Lamare, D. A. and Malissen; Carrier, A.; Guerder, S. Thymus-specific serine protease controls autoreactive CD4 T cell development and autoimmune diabetes in mice. Journal Article In: J Clin Invest , vol. 121, no. 5, pp. 1810-1821, 2011. @article{Viret2011,
title = { Thymus-specific serine protease controls autoreactive CD4 T cell development and autoimmune diabetes in mice.},
author = {Viret, C. and Leung-Theung-Long, S. and Serre, L. and Lamare, C.and Vignali, D.A.and Malissen, B. and Carrier, A. and Guerder, S. },
url = {https://www.ncbi.nlm.nih.gov/pubmed/?term=Thymus-specific+serine+protease+controls+autoreactive+CD4+T+cell+development+and+autoimmune+diabetes+in+mice.},
doi = { 10.1172/JCI43314},
year = {2011},
date = {2011-05-01},
journal = {J Clin Invest },
volume = {121},
number = {5},
pages = {1810-1821},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
Viret, C.; Lamare, C.; Guiraud, M.; Fazilleau, N.; Bour, A.; Malissen, B.; Carrier, A.; Guerder, S. Thymus-specific serine protease contributes to the diversification of the functional endogenous CD4 T cell receptor repertoire. Journal Article In: J Exp Med., vol. 208, pp. 3-11, 2011. @article{Viret2011b,
title = {Thymus-specific serine protease contributes to the diversification of the functional endogenous CD4 T cell receptor repertoire. },
author = {Viret, C. and Lamare, C. and Guiraud, M. and Fazilleau, N. and Bour, A. and Malissen, B. and Carrier, A. and Guerder, S. },
url = {https://www.ncbi.nlm.nih.gov/pubmed/21173102},
doi = {10.1084/jem.20100027},
year = {2011},
date = {2011-01-17},
journal = {J Exp Med.},
volume = {208},
pages = {3-11},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
|
 Using specific antigen models that we have developed in the lab, our objectives are to evaluate: 1- the nature of the delivery system and the nature of the immunomodulatory signals that control these events, 2- the links between the anatomy of the memory compartments and memory fitness, 3- the influence of vaccine formulation on Ag availability and persistence, 4- the crosstalk between different memory cells that tightly collaborate during recall responses.
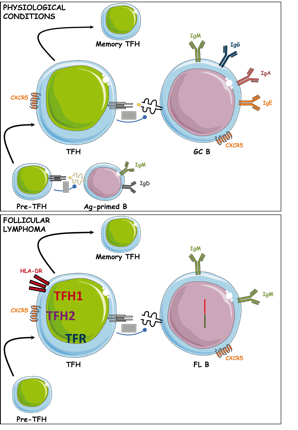
Using specific antigen models that we have developed in the lab, our objectives are to evaluate: 1- the nature of the delivery system and the nature of the immunomodulatory signals that control these events, 2- the links between the anatomy of the memory compartments and memory fitness, 3- the influence of vaccine formulation on Ag availability and persistence, 4- the crosstalk between different memory cells that tightly collaborate during recall responses. We now describe in depth memory Tfh cells and evaluate the physiological impact of these cell interactions on the quality and longevity of a humoral response in mouse and human settings. Moreover, we also study human autoimmune pathologies mediated by auto-antibodies such as Pemphigus vulgaris and assess how the response of patients to B-cell depleting treatment relies not only on the inhibition of the auto-reactive B cells but also on their cognate cell regulators, namely the Tfh cells.
We now describe in depth memory Tfh cells and evaluate the physiological impact of these cell interactions on the quality and longevity of a humoral response in mouse and human settings. Moreover, we also study human autoimmune pathologies mediated by auto-antibodies such as Pemphigus vulgaris and assess how the response of patients to B-cell depleting treatment relies not only on the inhibition of the auto-reactive B cells but also on their cognate cell regulators, namely the Tfh cells.